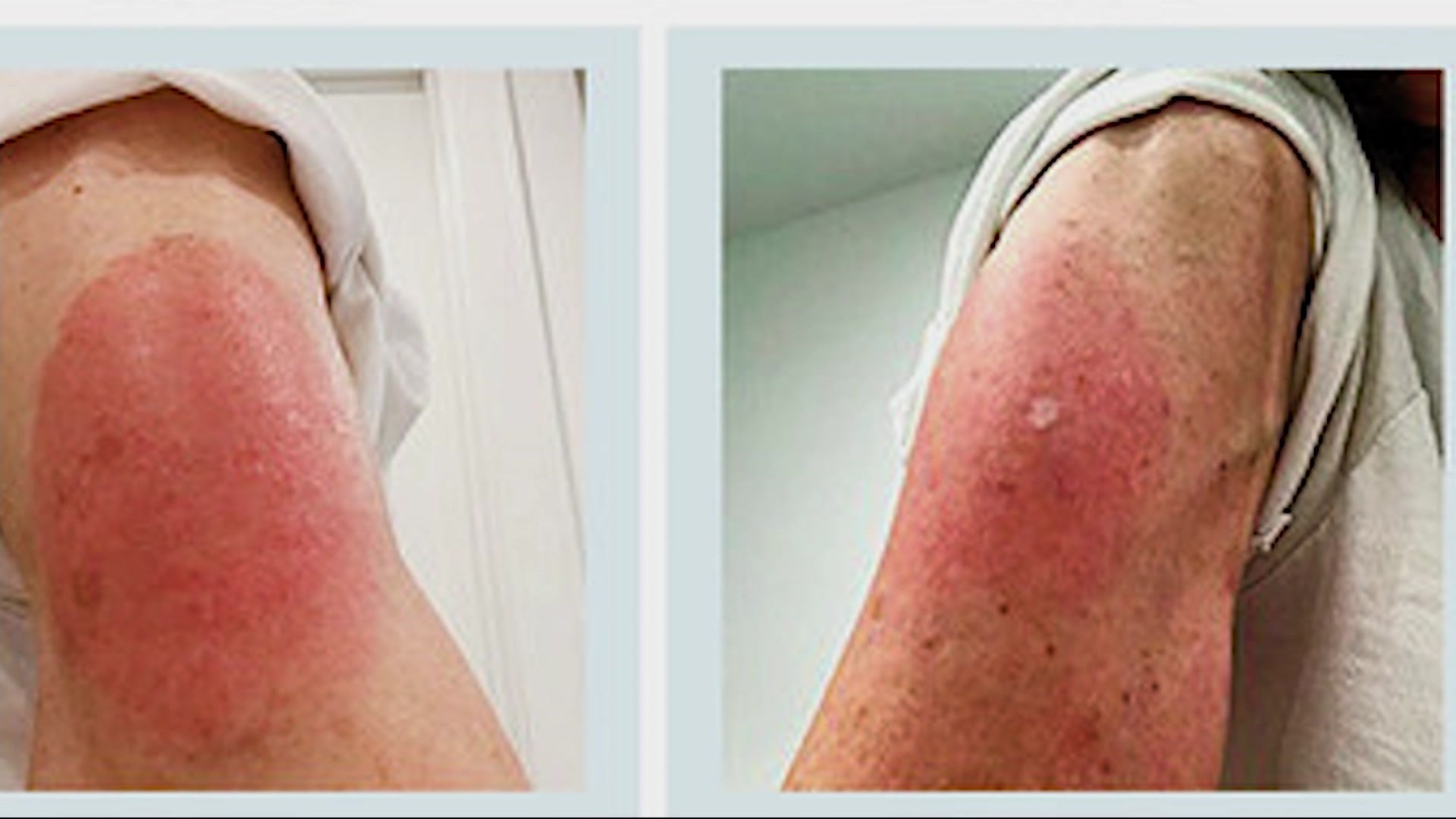
In this study, the majority of the participants were aged 40 years and above (73%). Women may, therefore, be counselled before the administration of vaccines so that they would be less worried about the higher level of side effects that they might experience after vaccination. This might be a plausible explanation why women experienced fatigue, headache and drowsiness more than males in the current study. It was reported in several studies that testosterone is related to immune response suppressive action in adult males, while estradiol stimulates a higher antibody response to viral infections in adult females. Most likely, the differences in immune responses are associated with variations in male and female hormones. Several factors might be responsible for this gender disparity in vaccine side effects. In another study, it was stated that women (20–59 years old) were four times more likely than men to report post-vaccination allergic responses following the 2009–10 H1N1 pandemic vaccine administration. For instance, a study describing the reports within VAERS (Vaccine Adverse Event Reporting System) of the CDC from 1990 to 2016 mentioned that 80% of the anaphylaxis reports were from female participants. However, this is similar to the trend displayed by adverse vaccine event monitoring studies of Pfizer-BioNTech vaccine in Saudi Arabia and other vaccines in general. This pattern was not particularly reported in the clinical trials of Oxford/AstraZeneca. The study revealed that a significantly greater number of female participants suffered from post-COVID-19 vaccination side effects compared to males ( p = 0.016). This might have happened because of differences in ethnicity, geographical location and environmental factors of the study population. The most commonly reported symptoms in this investigation were pain at the site of injection (48.9%), feeling feverish (28.3%), fever (24.3%), myalgia (23.2%), fatigue (17.5%) and headache (13.7%) ( Figure 4), which are consistent with the clinical trial results of Oxford/AstraZeneca’s ChAdOx1 nCoV-19, although the frequencies of the side effects were much lower in this study. These common side effects are short-lived and much less serious than developing COVID-19 or complications associated with COVID-19. After a vaccine administration, some side effects usually occur, which indicates that the vaccine is activating the body’s immune system to defend itself from the disease. Hence, research publications and information regarding the safety and efficacy of these vaccines are highly called for to prevent vaccine-related misconception and promote uptake of available vaccines. :max_bytes(150000):strip_icc()/when-to-expect-covid-19-vaccination-side-effects-5176621_V1-67a15605548f4e64b078cf5ab85213d1.jpg)
The unprecedented pace at which COVID-19 vaccines were developed had heightened the already existing vaccine hesitancy among general population, and further aggravated by the unregulated circulation of conspiracy theories and misinformation in the social media. Nevertheless, further long-term follow-up study with a larger sample size is warranted to establish the long-term safety of the COVID-19 vaccine. 
It was concluded that the Covishield vaccine was well-tolerated among people of different age groups. 
In addition, a significantly higher percentage of female participants suffered from post-vaccination side effects compared to males (OR = 1.51). Logistic regression analysis further revealed that compared to people aged 70 years or above, the incidence of reported side effects was significantly higher in people aged 18–30 years (odds ratio (OR) = 8.56), 31–40 years, (OR = 5.05), 41–50 years (OR = 4.08), 51–60 years (OR = 3.77) and 61–70 years (OR = 3.67). These findings were consistent with the results indicated by the clinical trial of ChAdOx1nCoV-19. Pain at the site of injection, fever, myalgia, fatigue and headache were the most commonly reported symptoms, and the overall side effects were found to be significantly more prevalent in the younger population ( p ≤ 0.05). The results included the responses of 474 vaccine recipients from March–April 2021. The study was conducted using both online and printed questionnaires and the data were analysed using SPSS. The purpose of this study was to evaluate the side effects that were experienced by the Bangladeshi residents after receiving the first dose of the Oxford-AstraZeneca’s Covishield vaccine (ChAdOx1nCoV-19). In response to the raging COVID-19 pandemic, Bangladesh started its vaccine administration in early 2021 however, due to the rapid development and launch of the vaccines in the market, many people had concerns regarding the safety of these vaccines.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
